EMERGING FOOD-BORNE PATHOGENS
In this issue:
• Introduction
• Enterobacter sakazakii
• Listeria monocytogenes
• Escherichia coli
• Campylobacter spp.
• Epilogue
• News Section
1. INTRODUCTION
• In December 2004 in France, the infant formula Pregestimil, a specialist milk formula for premature infants, was implicated in the serious illnesses of 9 infants resulting in 2 deaths. Investigations revealed that 4 implicated lots of the product produced over a period of 6-months were contaminated with Enterobacter sakazakii at levels ranging from 1 to 10 cfu per 100g (1, 2).
• 5 cases of infants with E. sakazakii infections resulted in one death due to meningitis in New Zealand in 2004. The infections were linked to powdered infant formula used in a nursery (1, 2).
• An outbreak of E. sakazakii infection in 12 infants was noticed in a hospital in Belgium during June-July 1998. At least 3 patients were infected with isolates that were similar to the one found in infant formula. The infections resulted in death of 2 infants (1).
• In another incident in France in 1994, an outbreak of E. sakazakii infection in 18 infants was linked to a consumption of contaminated infant formula which resulted in 4 deaths (1).
• Consumption of milk, contaminated with Listeria monocytogenes, from the Whittier Farms plant in Shrewsbury fatally sickened 3 elderly men in USA (Massachusetts) during the period June 2007–January 2008. The milk also sickened 2 women, one of which was pregnant and then miscarried. The deadly strain of the bacterium was found in 7 bottles and on the floor near the homogenizer at the Shrewsbury plant (3).
• Two cases of infection in children in Grey Bruce were linked to raw milk products, as reported by the public health officials in March 2007 in Canada. Of these, one child was taken to hospital with yersiniosis, a gastrointestinal infection, after drinking raw milk on her family farm. Another infant was confirmed to have listeriosis, an infection caused by L. monocytogenes, which seems to have come from raw milk cheese consumed by the baby's mother (3).
• In January 2007 in Czech Republic, health officials said 13 people had died of listeriosis (L. monocytogenes infection) and more than 90 people had fallen ill with Listeria infection during the past several months. The strain of bacteria had been found to be present in soft cheese and some other non-dairy foods (3).
• In UK in 1999, 3 persons were taken ill after consuming cheese made from unpasteurized milk. The cheese was contaminated with E. coli (VTEC O157) (4).
• In yet another incident in UK in 1998, consumption of unpasteurized cream contaminated with E. coli (VTEC O157) caused illness in seven persons (4).
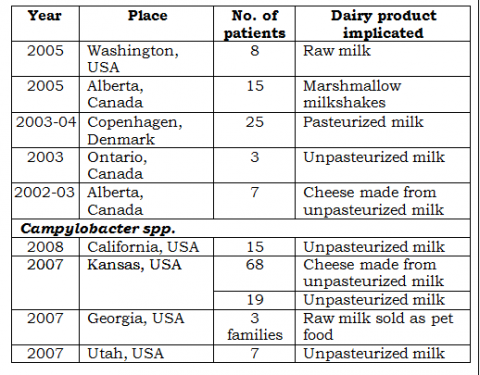
• See the following table.
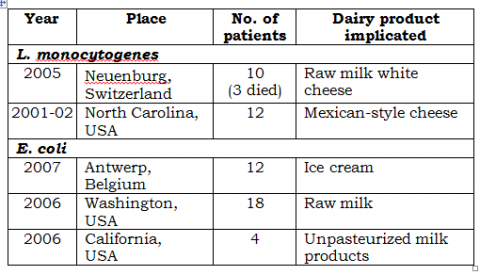
These are some of the reported cases of outbreaks of food-borne illness caused by some emerging pathogens.
In recent years, the epidemiology of food-borne diseases has been changing with the emergence of new pathogens as well as increased incidence of illnesses caused by several known pathogens in the recent past. New food-borne diseases occur for a number of reasons which include increase in international trade resulting in introduction of pathogens into new areas; microbial adaptation; and changes in food production system, human populations and lifestyles.
Food-borne diseases that are regarded as emerging include illnesses caused by Enterobacter sakazakii, Listeria monocytogenes, enterohaemorrhagic Escherichia coli [EHEC / VTEC (Vero toxigenic), particularly serovar O157:H7], Campylobacter spp., etc.
Details of various common pathogens found in milk and milk products were provided in previous issues of Technews (No.31, March-April 2001; and No.32 May-June 2001). This issue of Technews provides a brief insight of some of the above-mentioned emerging pathogenic microorganisms as a result of their presence
in various types of milk products and hence a potential source of infection.
2. Enterobacter sakazakii (1, 2, 4-8)
E. sakazakii is a gram-negative, non-sporeforming bacterium belonging to the Enterobacteriaceae family, which contains a number of bacterial species found in the human and animal gut and the environment. It qualifies as a coliform bacterium.
It is an opportunistic pathogen emerging as a public health concern. It has been associated with a number of outbreaks of infection, usually meningitis, in premature neonates, with high rates of morbidity and mortality. In the few outbreaks reported,
20% to >50% of the infants who contracted the disease died. For survivors, severe lasting complications resulted including neurological disorders. The outcome related to adult disease seems to be significantly milder.
2.1 Groups at risk: Enterobacter sakazakii has caused disease in all age groups. From the age distribution of reported cases it is deduced that infants (children less than 1 year old) are at particular risk. Among infants those at greatest risk for Enterobacter sakazakii infection are neonates (up to 4 weeks of age), particularly pre-term infants, low birth weight infants or immuno-compromised infants.
The true magnitude of the E. sakazakii infections is unknown due to lack of surveillance and reporting systems in most countries. The frequency of the disease in infants appears to be very low, yet the disease is devastating. A review of cases in infants reported in the English literature from 1961 to 2003 found 48 cases of Enterobacter sakazakii induced illness among infants. The US FoodNet 2002 survey found that the rate of invasive Enterobacter sakazakii infection among infants under one year old was 1 per
100,000. Significant long-term effects in the form of neurological deficiencies can result from the infection, especially among those with severe meningitis and cerebritis.
2.2 Implicated foods: Although E. sakazakii can be present in any food article, its presence in powdered infant formula, even at very low levels, is of significance. Contaminated powdered infant formula has been convincingly shown, both epidemiologically and microbiologically, to be the vehicle and source of infection in infants.
The organism has been reported to be isolated in powdered formula production environments, other food factories as well as in households.
2.3 Sources of contamination: The presence of E. sakazakii in powdered infant formula can occur as a result of:
• Contamination through ingredients not submitted to heat treatment during the powdered infant formula manufacturing process
• Contamination from the processing environment during the dry steps in the manufacturing process.
2.4 Control of E. sakazakii in powdered infant formula: Quality of raw materials, air and liquid filters, sifter screens, magnets/metal detectors, pasteurization and storage temperatures are important control points and must be addressed specifically.
• Control of ingredient quality: In order to ensure that ingredients are microbiologically suitable, a number of factors need to be considered:
− The likelihood of occurrence in ingredients: some ingredients are considered to have a high risk of containing Enterobacteriaceae (e.g. starch) while others have a low risk (e.g. oils)
− Selection of the ingredient supplier according to stringent criteria (e.g. appropriate control measures, good hygienic practices, verification and release procedures in place)
− Testing of ingredients to verify effectiveness of the above measures
• Processing: A high thermal resistance of E. sakazakii strains in comparison to other members of the Enterobacteriaceae was believed to be responsible for high prevalence in powdered infant formula. However, recent studies suggest that the osmo- tolerance of the organism may be more important in this regard. The ability to be osmo-tolerant may increase the risk of the organism becoming more dominant in the environment, thus increasing the risk of post-process contamination. Various studies have revealed that substantial diversity in thermal resistance is experienced among strains, yet inactivation of the organism occurs very quickly at temperatures above 70ºC.
Post processing inactivation of microbial pathogens in powdered formula in packaged form seems possible only by using irradiation. However, with doses that are likely to be
required to inactivate E. sakazakii in the dry state, the technology does not appear to be feasible due to organoleptic deterioration of the product. A number of other technologies, such as ultra-high pressure and magnetic fields, may be potential alternatives, but these technologies are at an early stage of development and currently none is suitable for dried foods.
• At final consumption stage: Caregivers should include a decontamination step in the preparation of powdered infant formula (such as reconstituting with boiling water or heating reconstituted formula).
A preliminary risk assessment further indicated that reduced holding (time between rehydration of the formula and consumption) and feeding times for the reconstituted formula decrease the risk of infecting infants. A combination of control measures would have the greatest impact on reducing the risk.
A detailed description of various hygienic practices in the production and handling of the powdered infant formula and similar other products are provided in the recently developed Codex Code of Hygienic Practice for Powdered Formulae for Infants and Young Children (CAC/RCP 66-2008).
3. Listeria monocytogenes (4-6, 9)
L. monocytogenes is the main human pathogen among the six distinguishable species under the Listeria genus.
The organism has been recognized as a significant food-borne pathogen only since the early 1980s when outbreaks of food- borne listeriosis demonstrated the severe nature of the illness with exceptionally high levels of mortality, particularly in the most vulnerable members of the community such as unborn babies, the elderly and the immuno-compromised.
The symptoms can take a few days or even weeks to appear and may include fever, chills, muscle aches, diarrhoea or upset stomach, headache, stiff neck, confusion, and loss of balance. In more serious cases, listeriosis could also lead to death.
3.1 Groups at risk: Pregnant women are about 20 times more likely to get listeriosis than other healthy adults, and an estimated
⅓rd of all listeria cases occur in pregnant women. Pregnant women who are infected with listeriosis also can pass the infection to their unborn babies without even knowing it. During the 1st trimester of pregnancy, listeriosis may cause miscarriage. As the pregnancy progresses to the 3rd trimester, the mother is more at risk. Listeriosis can also lead to premature labour, the delivery of a low-birth-weight infant, or infant death. Foetuses which suffer a late infection may develop a wide range of health problems, including mental retardation, paralysis, seizures, blindness, or impairments of the brain, heart, or kidney. In newborns, L. monocytogenes can cause blood infections and meningitis.
3.2 Implicated foods: The organism in widely distributed in the environment and occurs in almost all food raw materials from time to time. Food-borne listeriosis has been attributed to contaminated ready-to-eat meats, poultry, seafood, and dairy products, including soft ripened cheeses and products made from unpasteurized milk. Raw milk is a well-recognized source of Listeria.
Listeria contamination of processed dairy products is most likely attributed to post-pasteurization contamination from the dairy
plant environment i.e. coolers, freezers, processing rooms, entrances, tray washers, floor mats, foot baths etc.
Due to its capacity to grow at very low temperatures (as low as - 0.4°C) and colonize factory environments (moist/wet environments; biofilms on process equipment), the organism has been able to exploit chilled, ready-to-eat food products that are intended to have long shelf life under refrigerated conditions.
3.3 Factors responsible for increased risk of food-borne listeriosis: The factors that appear to elevate the risk of listeriosis due to contamination and growth of L. monocytogenes in foods include the following:
• Raw material or product exposed to contamination
• Product manufactured with no processing stage capable of destroying the organism, e.g. cooking
• Product with little or no preservation factors e.g. neutral pH, low salt, high moisture
• Product exposed to post pasteurization contamination
• Product sold with long shelf-life under chilled conditions
• Improper handling of ready-to-eat products.
3.4 Control of Listeria monocytogenes in foods: Control of
Listeria spp. in foods is dependent on four key factors:
• Preventing contamination of raw materials or growth in raw materials, if present - L. monocytogenes is a relatively frequent contaminant of raw milk. Once introduced into the dairy environment the organism readily colonizes moist environments. The capacity to grow at low temperatures results in increase in the bacterial numbers even during refrigeration. It is therefore essential to ensure that cleaning and disinfection of the environment and raw milk handling equipment are carried out properly. Failure to do so will inevitably lead to increased incidence and levels of these organisms in the raw milk.
• Destroying or reducing the organism during processing - Heat treatment is often used as a primary process to destroy Listeria organism in the manufacture of a variety of food products, including dairy desserts. Pasteurized milk, heat treated at 71.7°C for 15 seconds, is sufficient to achieve a 3-4 log reduction in the organism.
• Preventing recontamination from the factory environment, equipment or personnel after a reduction or destruction stage during processing - The measures include:
- Separation of raw material processing (high risk) area and finished product (low risk area)
- Effective cleaning and disinfection of the environment and equipment to prevent the organism from building up and presenting a hazard to the product.
- Cleaning of other non-food contact surfaces (potential ‘reservoirs’) such as table ledges, door handles, overhead pipes, chillers, floors, walls etc.
- Monitoring of cleaning efficiency, such as conducting ATP bioluminescence tests and effectively targeted tests for L. monocytogenes.
• Minimizing its growth, if present, during the shelf life of the final product - Being not always possible to exclude contamination completely, the organism will be an occasional contaminant of refrigerated products such as ripened cheeses. The ultimate safety of these products rests on ensuring high levels of the organisms are not allowed to develop in the product during its shelf life.
3.5 Preventing food-borne listeriosis: Listeriosis can also be prevented through the use of following measures:
- Refrigerate or freeze perishables, prepared food, and leftovers within 2 hours of eating or preparation. Follow the 2-Hour Rule: Discard food that has been left out at room temperature for longer than 2 hours. When temperatures are above 32ºC, discard food after one hour.
- Use ready-to-eat, perishable foods, such as dairy, meat, poultry, seafood, and produce, as soon as possible.
- Raw (unpasteurized) milk or dairy products prepared from or containing unpasteurized milk should be avoided.
A detailed description of various measures for control of L. monocytogenes in ready-to-eat foods is provided in the recently developed Codex Guidelines on the Application of General Principles of Food Hygiene to the Control of Listeria monocytogenes in Ready-to-Eat Foods (CAC/GL 61-2007).
4. Escherichia coli (4-6, 9, 10)
E. coli is a common and harmless member of the commensal microflora of the intestinal tract of humans and other warm blooded animals. However, it is only in more recent years that the food industry has been confronted with some strains of E. coli as a cause of significant morbidity and mortality in outbreaks of food-borne infections, in particular those caused by the Vero toxin (also referred to as Shiga toxin) producing E. coli (VTEC or STEC). Many of these organisms fall under the category of Entero-haemorrhagic E. coli (EHEC) and all EHEC are considered human pathogens.
E. coli 0157:H7 is currently the most predominant food-borne VTEC. However, other strains like O26, O103, O111, O118 and O145 are increasingly being recognized as posing an equal or possibly greater threat to human health.
Illnesses caused by VTEC E. coli include haemorrhagic colitis (sudden onset of severe crampy abdominal pain, grossly bloody diarrhoea and vomiting), haemolytic uraemic syndrome (bloody diarrhoea, acute renal failure in children, acute nephropathy, seizures, coma, death), thrombotic thrombocytopaenic purpura (similar to above but also fever, central nervous system disorders, abdominal pain, gastrointestinal haemorrhage, blood clots in brain, death).
4.1 Implicated foods: A wide variety of foods have been implicated in outbreaks of illness attributed to VTEC E. coli. Among dairy products, these include French brie cheese, Camembert cheese, yoghurt, unpasteurized cream, cheese made from unpasteurized milk etc.
4.2 Factors responsible for increased risk of food-borne E. coli infections: Factors likely to be associated with increased risk of E. coli infection include all of the following:
• Raw material or product exposed to contamination from bovine origin (faeces)
• Product manufactured with no processing stage capable of destroying the organism, e.g. cooking
• Product exposed to post pasteurization contamination
• Improperly handled ready-to-eat products
• Contact with an infected individual or animals
4.3 Control of E. coli in foods: Control of E. coli in food production is achieved through the application of effective control of raw materials, process conditions, post-process contamination etc.
• Raw material control: The primary route of contamination of raw material (milk) is from exposure to animal faeces, from bovine source in particular, which contaminate the udder of the animal. Control measures could include:
- Effective milking hygiene which includes cleaning and disinfection of udder and other milking equipment and vessels
- Routine monitoring of raw milk for indicators of contamination including E. coli.
• Control in processing: Heat processing is probably the effective means of eliminating almost all types of E. coli from the food products, including dairy products such as pasteurized milk, cheese, yoghurt. Pasteurization of milk at 71.7ºC for 15 seconds is considered sufficient to reduce the contamination levels by more than 6 log cfu/g.
• Post process controls: Like any other bacterial pathogen a recurring factor often identified as a reason for outbreaks of VTEC E. coli infections is the contamination of the product after application of the pathogen reduction process. Outbreaks implicating pasteurized milk and cheese have indicated post process contamination (through raw material or environment) as a source of this organism in the implicated products.
Segregation of low and high risk areas in the production line to avoid cross contamination; effective cleaning and sanitation of all equipment to prevent the organism from building up in the process equipment; and implementation of a efficient employee hygiene policy are important measures for preventing post process contamination of the products.
5. Campylobacter spp. (4-6)
Campylobacter spp. are gram-negative, micro-aerophilic, non- spore forming bacteria and are part of the normal intestinal microflora of a wide variety of domestic and wild animals. These are increasingly implicated in bacterial diarrhoea in humans. Among various species, C. jejuni is regarded as the leading cause of bacterial food-borne infection and is responsible for 80-90% of the campylobacteriosis. Other less prevalent species include C. coli, C. upsaliensis and C. lari.
Campylobacteriosis in humans is usually characterized by an acute, self-limiting entero-colitis with symptoms such as abdominal pain and cramps, and diarrhea which may be inflammatory (with slimy/bloody stools) or non-inflammatory (with watery stools and no blood). Infection with C. jejuni is also associated with Guillain–Barre syndrome (GBS), an autoimmune peripheral neuropathy causing limb weakness that is sometimes fatal.
Foods associated illness usually results from eating foods of animal origin that are raw or inadequately heat treated or eating foods that are recontaminated after processing. Milk is an important and frequent source in the transmission of Campylobacter enteritis. C. jejuni is a known cause of bovine mastitis and consumption of unpasteurized milk from affected animals leads to gastroenteritis in people. Most of the milk-borne Campylobacter enteritis have been associated with the consumption of unpasteurized milk. Some other reports, however, indicate that Campylobacters have not been associated with dairy products such as raw milk cheeses.
The virulence of the organism (relatively low infectious dose of a few hundred cells) and its widespread prevalence in animals are factors for it being a leading cause of gastroenteritis in people.
However, the organism is susceptible to environmental conditions (high temperature, dry environments and presence of oxygen) and does not survive well in foods. Hence, it is relatively easy to control. Campylobacter is relatively heat sensitive and normal heat processes would adequately facilitate control of the pathogen. A cleaning procedure using hypochlorite in addition to the cleaning with hot water is effective in reducing the level of Campylobacter contamination.
6. EPILOGUE
The emerging food-borne infections have gained significant importance due to their increasing incidence and resultant morbidity and mortality. The capability of these organisms to cause illness in people that consume products (even with low level of contamination) poses a serious challenge to the food manufacturers, processors and handlers in ensuring a contamination free product. Yet, the difficulties could be successfully overcome by implementing an effective food safety system and maintaining strict hygienic conditions in the production and processing environment.
REFERENCES
1. WHO & FAO (2008), Microbiological Risk Assessment Series: Enterobacter sakazakii (Cronobacter spp.) in powdered follow- up formulae (Meeting Report), FAO, Rome and WHO, Geneva.
2. WHO & FAO (2006), Microbiological Risk Assessment Series
10: Enterobacter sakazakii and Salmonella in powdered infant formula (Meeting Report), FAO, Rome and WHO, Geneva.
3. International Society for Infectious Diseases (ISID): ProMED- mail, the Program for Monitoring Emerging Diseases- (Website- http://www.promedmail.org)
4. Blackburn. C. de W., McClure, J. (2002), Food-borne Pathogens: Hazards, risk analysis and control, Woodhead Publishing Limited, Cambridge, England
5. Hui, Y.H., Pierson, M.D. and Gorham, J.R. (2001), Food-borne Diseases Handbook- Volume 1: Bacterial Pathogens, Marcel Dekker Inc., New York.
6. Jay, J.M. (2000), Modern Food Microbiology (Sixth Edition),
Aspen Publishers Inc., Maryland
7. WHO & FAO (2004), Microbiological Risk Assessment Series 6:
Enterobacter sakazakii and other microorganisms in powdered infant formula (Meeting Report), FAO, Rome and WHO, Geneva.
8. Erickson, M.C. and Kornacki J.L., Enterobacter sakazakii: An Emerging Food Pathogen, University of Georgia, Center for Food Safety, Griffin, GA (Website- http://www.ugacfs.org/faculty/ Erickson/EBWhitePaper.mpd.pdf)
9. WHO (2002), Food-borne diseases, emerging (Fact Sheet NO 124), World Health Organization, Geneva
10. Gyles, C.L. (2006), Shiga toxin-producing Escherichia coli: An overview, Journal of Animal Science, 2007.85:E45-E62
* * *
NEWS SECTION Indian Food Laws
• Notification GSR 591(E) of 13 August 2008 of the Ministry of Health and Family Welfare: The notification is a latest
corrigendum to an earlier notification GSR 356(E) of 7 June
2005 (Refer Technews Issues No.60, January–February 2006). The Corrigendum indicates that the provisions in the GSR 356 (E) on the definitions, food additive provisions and microbiological specifications for specified dairy products (cheese, processed cheese, processed cheese spread, ice cream/
kulfi/chocolate ice cream/softy ice cream, dried ice cream mix/
dried frozen dessert/confection, frozen dessert/frozen confection, milk ice/milk lolly and yoghurt) shall come into force after 3 years and 6 months from the date of publication of GSR 356(E). Therefore, these provisions are now likely to be applicable from 7 December 2008.
• Notification GSR 664 (E) of 19 September 2008 of the Ministry of Health and Family Welfare: The notification amends PFA rules, as amended earlier by the GSR 491 (E) dated 21 August 2006 (now applicable from 18 March 2009 as indicated below). The Rules 32 (labelling requirements), 37 A (manufacture of proprietary foods and 42 (form of labels – declaration of allergenic ingredients) have been further revised. The revised Rules are scheduled to become effective from 18 March 2009.
The important revision in Rule 32 is the requirement to specify the quantity of sugar when declaring amount of carbohydrates.
Rule 37 A has been revised to remove the requirement to obtain a separate license for manufacture of proprietary products.
Rule 42 is has been revised to remove the requirement to declare the presence of allergenic ingredients in foods.
• Notification GSR 730 (E) of 13 October 2008 of the Ministry of Health and Family Welfare: It is a latest corrigendum to the GSR 491 (E) of 21 August 2006 of the Ministry of Health and family Welfare that amended the PFA Rules by defining several new terms, revising Rules 32 (labelling requirements), 37A (proprietary foods), 37B (infant milk substitute/infant foods) and 42 (form of labels) (refer Technews issue 64, September-October 2006). The Corrigendum indicates that the provisions of GSR 491 (E) would now be applicable from 18
March 2009.
Codex Alimentarius Commission (CAC)
The period December 2008-February 2009 features meeting of the Codex Committee on Food Hygiene during 1-5 December
2008 at Guatemala City, Guatemala. The important issues that would be discussed are establishing of microbiological criteria for follow-up formulae and for Listeria monocytogenes in ready-to- eat foods.
International Dairy Federation (IDF)
IDF has published the following Bulletin/Guide/Standards recently:
• IDF Bulletin No.432/2008: The World Dairy Situation 2008;
• Guide to Good Animal Welfare in Dairy Production 2008 (This IDF publication is available free of cost from the IDF Internet homepage: http://www.fil-idf.org/WebsiteDocuments/Guide to Good Animal Welfare in Dairy Production.pdf)
• IDF 9/ISO 1736: Dried milk and dried milk products - Determination of fat content - Gravimetric method (Reference method)
• IDF 13/ISO 1737: Evaporated milk and sweetened condensed milk - Determination of fat content - Gravimetric method
(Reference method)
• IDF 16/ISO 2450: Cream - Determination of fat content - Gravimetric method (Reference method)
• IDF 22/ISO 7208: Skimmed milk, whey and buttermilk - Determination of fat content - Gravimetric method (Reference method)
• IDF 50/ISO 707: Milk and Milk Products - Guidance on Sampling
• IDF 59/ISO 1854: Whey cheese - Determination of fat content - Gravimetric method (Reference method)
• IDF 105/ISO 488: Milk - Determination of fat content – Gerber butyrometers
• IDF 116/ISO 7328: Milk-based edible ices and ice mixes - Determination of fat content - Gravimetric method (Reference method)
• IDF 123/ISO 8381 Milk-based infant foods - Determination of fat content - Gravimetric method (Reference method)
• IDF 197/ISO 20541: Milk and milk products - Determination of
nitrate content - Method by enzymatic reduction and molecular- absorption spectrometry after Griess reaction
• IDF 226/ISO 2446: Milk - Determination of fat content